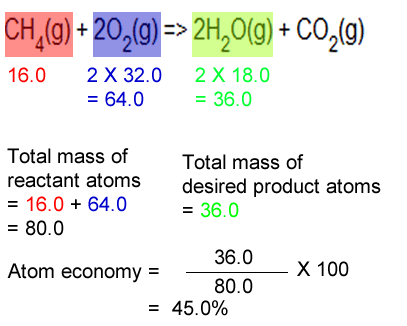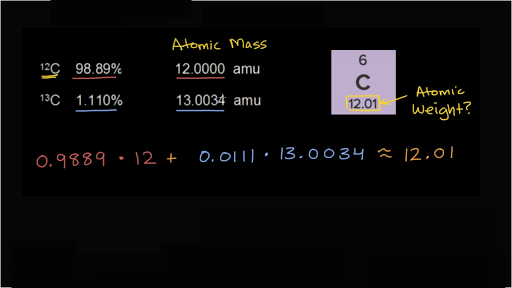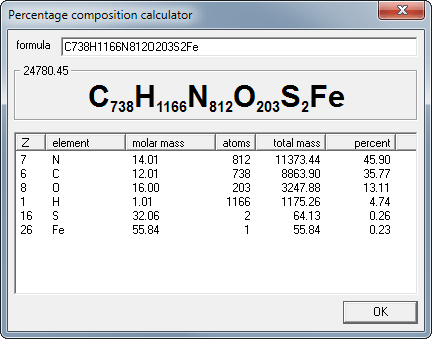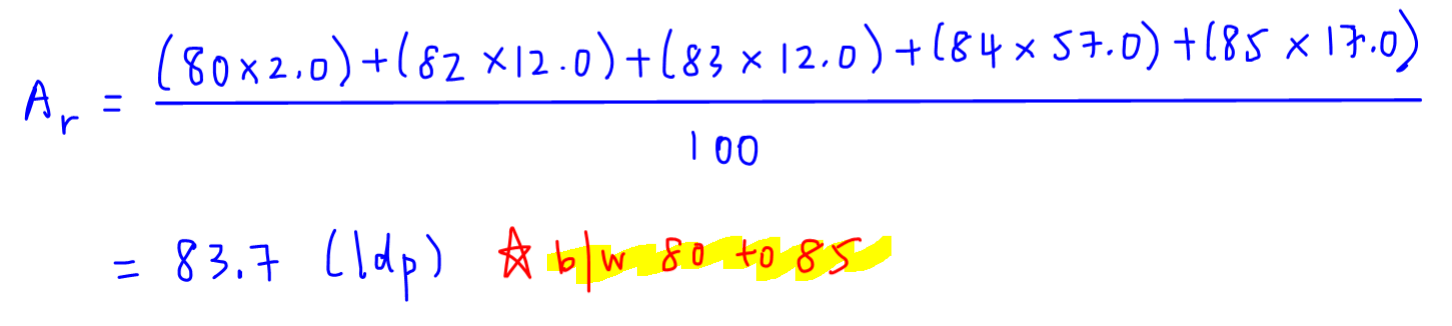
How to Calculate and Solve for Conversion of Weight Percent to Atom Percent | Imperfection in Solids
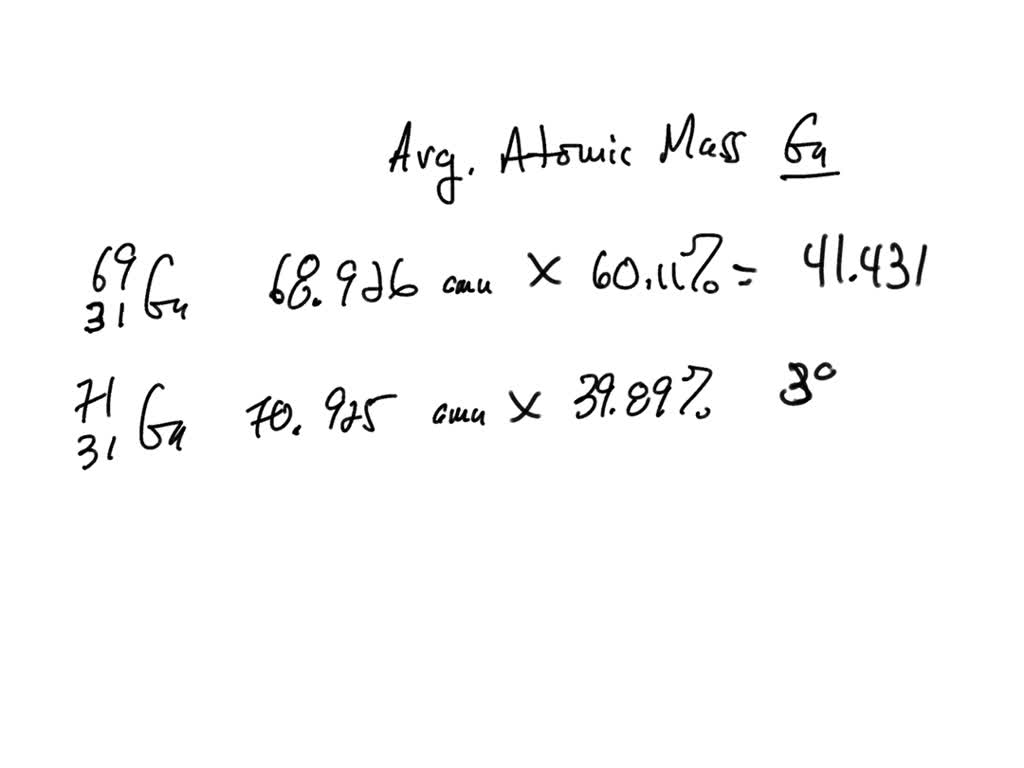
SOLVED: In nature, gallium (Ga) is found as two isotopes. Calculate the atomic mass of this element. 69 31Ga (atomic mass 68.926 amu, abundance 60.11%) 71 31Ga (atomic mass 70.925 amu, abundance 39.89%)









:max_bytes(150000):strip_icc()/mass-percent-composition-example-609567_V2-01-89c18a9d30ea43b494d09b81f7ffefc1.png)