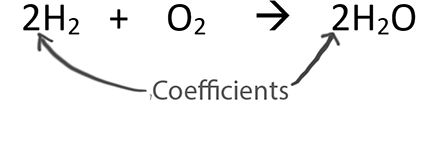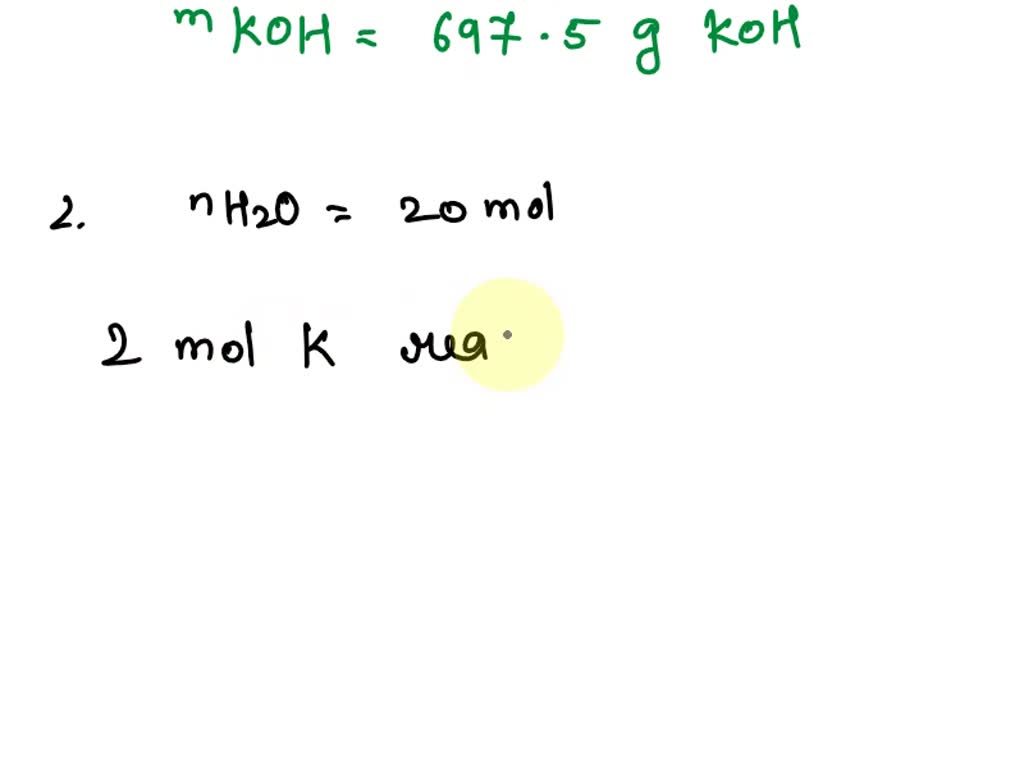
SOLVED: K + H2O –> KOH + H2 1. How many grams of KOH are produced if 224 g of H2O are used? 2. How many moles of K are used if

SOLVED: Are these equations balanced or unbalanced? H2 + O2 > H2O H2 + O2 > H2O2 K + O2 > K2O Na + Cl > NaCl

SOLVED: Write a balanced net ionic equation for the reaction of potassium with water. a. 2 K(s) + H2O(l) â†' No reaction b. 2 K(s) + 2 H2O(l) â†' 2 K+(aq) +

Chemistry 2/10/14 “Mystery creates wonder and wonder is the basis of man's desire to understand.” –Neil Armstrong. - ppt download

SOLVED: Balance the following equations. Do not include the states of matter. (a) C + O2 -> CO2 (b) KOH + H3PO4 -> K2HPO4 + H2O (c) N2 + H2 -> NH3 (d) K + H2O -> KOH + H2

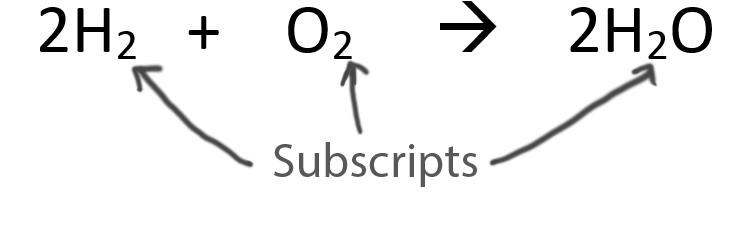





![Punjabi] Balance the chemical equation : K + H2O → KOH + H2 Punjabi] Balance the chemical equation : K + H2O → KOH + H2](https://static.doubtnut.com/ss/web-overlay-thumb/10301142.webp)